What is the retrobrite chemical reaction?
Created
Modified
Published
Introduction
Some plastics yellow over time, especially parts exposed to air and light. In the retro-computing community, it is well-known that this yellowing can often be reversed with a UV and hydrogen peroxide reaction, sometimes called retrobright or retrobrite (stylized as retr0bright). [1] I am surprised that this process works, because it sounds counterintuitive. On the one hand, the yellowing reaction is said to be caused by oxygen, heat, and UV rays. On the other hand, the reaction that reverses yellowing is also said to be caused by oxygen, heat, and UV rays. (Granted, it’s generally done in solution rather than open air, but even this is not always needed, as we will see below.)
Contrast this with the oxidation of iron: rust forms in the presence of oxygen and moisture, and rusting is accelerated by hydrogen peroxide. Additional oxygen, moisture, and hydrogen peroxide will not transform rust back into metallic iron. Similarly, if the yellowing reaction in plastic were due to oxygen and UV exposure, we would expect peroxide and UV exposure to accelerate the yellowing, not reverse it. Yet clearly the retrobrite process works. But the blog posts and forums have conflicting ideas about how.
Hypothesis #1: Bromine
Several sources say that bromine is the cause of the yellowing, and the hydrogen peroxide reacts with the bromine, reversing the yellowing:
Why do plastics yellow?
Manufacturers add bromine to ABS plastic to act as flame retardant. Over time, those chemicals react to the plastic’s basic polymers and turn yellow. Sunlight and Heat accelerate this process.
How is it Reversed?
The process uses peroxide and UV light (either a UVA bulb, or simple sunlight) to activate the reaction. This causes hydrogen to bond with bromine free radicals and restore the color.
http://www.retrofixes.com/2013/10/how-to-clean-whiten-yellowed-plastics.html
Now, in order to attach a Hydrogen atom to the Bromine to reduce it and make it stable, we need a source of it. This is where the Hydrogen Peroxide (H2O2) comes in. We want this in a solution of water so that we can break it apart.
https://web.archive.org/web/20130319002032/http://retr0bright.wikispaces.com/
The Vanish powder is something many people will have at home already. A chemical in the product called tetraacetylethylenediamine (TAED) acts as a catalyst in the reaction. TAED reacts with the hydrogen peroxide via a process called perhydrolysis, creating peracetic acid. This acid accelerates the removal of the yellow colour via epoxidation of the bromine.
https://devtty.io/2018/04/computing/no-nonsense-retrobrite-howto-guide/
The best explanation that I have for what we are doing is this. We are breaking apart hydrogen peroxide (H2O2) into O, H and water (H2O). Water dissociates into H and OH naturally. We are taking the oxygen away from the bromine using TAED and UV light, then replacing it with a hydrogen. The removed O links up with the O from the broken up peroxide and becomes O2 and is given off as bubbles. The picture below from Tonyyeb shows this crazy but useful mechanism at work; the bubbles are caused by the reaction on the surface of the ABS plastic.
https://web.archive.org/web/20120727044500/http://retr0bright.wikispaces.com/Catalyst
This is a process where more isn’t better; we are deliberately keeping the peroxide strength on the weaker side, so that the bromine molecules are favoured by the peroxide over the polymer; If you use very strong peroxide or add loads of Oxy, one of the side effects you might see is where the polymer gets attacked and turned to the hydroperoxide, usually by attacking the free butadiene bonds. What you will see is that the plastic takes on a white, ashen-looking “bloom” that unfortunately is permanent. This cannot be fixed and should be avoided.
https://web.archive.org/web/20121118042403/http://retr0bright.wikispaces.com/Problems+and+Pitfalls
If I’m interpreting this correctly, the proposed mechanism is that bromine is yellowish/brownish, so we need to remove the bromine from the plastic. Water and UV create hydronium ions (H3O+) that bond with the bromine ions (Br-), so the bromine is extracted from the surface of the plastic and goes into solution like this:
H3O+ + Br- → HBr + H2O
But if we wanted hydronium, we would use an acid like HCl, not an oxidizer like hydrogen peroxide (H2O2). Peroxides create hydroxide ions in solution (OH-), so if anything it will remove hydrogen from the plastic, not add to it. Oxygen has a higher electronegativity, so this reaction would tend to dominate:
OH- + HBr → Br- + H2O
But bromine, as we shall see soon, is a red herring; ABS will yellow all by itself, without any brominated fire retardant additives.
Hypothesis #2: Butadiene bleaching
It’s well-known for materials without any bromine can still yellow with age, including plastics like ABS. David Flood wrote a post in 2018 that highlights a specific mechanism for the butadiene degradation:
I’m sure we’ve all seen an old console or computer that’s all yellow and nasty. I’m also sure that you’ve heard of how it happens and how to get rid of it. While researching the entire yellowing and de-yellowing process, I came across a lot of misinformation and flat out lies. This article is a comprehensive study into both the entire process and clearing up false information that so often plagues the retro community.
[ … ]
The important part we will be looking at for how ABS yellows is that butadiene part. Quick organic chemistry: butadiene is incredibly reactive because the 4 carbons are all “conjugated”, which essentially means that the electrons have higher reactivity from being so close together. This component is the cause of ABS yellowing.
How ABS yellows
ABS yellowing is a well known radical based mechanism. A radical is any atom that only has an unpaired electron outside of its base state (electrons really love being in pairs). A mechanism is simply the process of a reaction. You don’t need to be knowledgeable in organic chemistry to understand this as its a fairly simple process
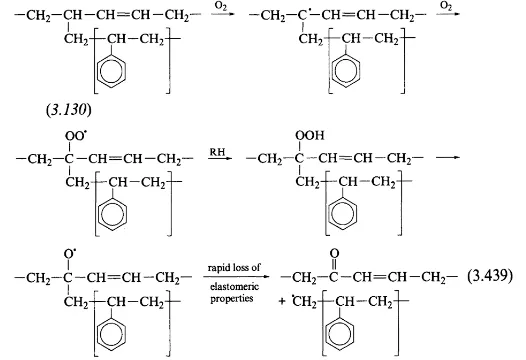
As you can see, only 2 things are involed, the carbon that is affected and simple oxygen. This process can be initiated by both heat and UV light, though UV increases the reaction rate by a much higher degree. And because this is radical based, there is no way to 100% stop it. It also ruins the mechanical strength of the plastic, making it much more brittle than fresh ABS.
[ … ]
There have been no real studies into how the deyellowing process works unfortunately, so what I say here is only from my knowledge in chemistry and what results I could find. The deyellowing process only requires hydrogen peroxide and a source of energy like UV light or heat. My guess is that it is simply the reverse reaction, but it does remove the pesky oxygen from the affected carbon. This entire process is enhanced by a peroxide bleaching agent like Tetra-acetylethylenediamine (TEAD). What is interesting is why this occurs, and in my research the plastic does not become deyellowed because of the hydrogen peroxide, but because of a bleaching agent.
He also links to a Quora post by Josh Velson which I will quote in full as Quora posts are easily lost to time:
This is a very interesting question.
The original method you are referring to, after some quick research, appears to have been developed by a group interested in plastics restoration and posted online in 2009 at http://retr0bright.wikispaces.com/ (now defunct).
On this basis, a lot of copycat tutorials have sprung up. Some examples include:
- Yellowing old consoles - The Solution.
- De-Yellowing plastic - the stain isn’t permanent after all - Creators Corner
These and many other sources around the web attribute this effect to the degradation of brominated polyols and other additives added to the acrylonitrile butadiene styrene (ABS) polymers as flame retardants. Examples of such flame-retardants can be found at http://www.tri-iso.com/icl-industrial.html In turn the earliest mention that I can find of this appears from http://Vintagecomputing.com and dates to 2007, in Why Super Nintendos Lose Their Color: Plastic Discoloration in Classic Machines, which publishes a reply from Nintendo that blames the brominated molecules for the discoloration of their consoles over time. This appears to be the main source of the attribution of discoloration to bromine.
I think it’s wrong.
This doesn’t jive with either my intuition, my professional experience, or my research. For one thing, bromides and bromates are generally colorless, or white; it’s only covalent bromine gas that is significantly colored. For another, ABS degradation is well studied. Under UV light, it begins an oxygen-limited photodegradation [1] (the same occurs in high heat conditions [2]) that results in yellowing. Notably, the oxygen-limited nature of photodegradation or thermal degradation is completely consistent with the virgin appearance of the inner parts of chipped old plastic despite discolored outsides. Thermal degradation also explains the yellowing in ABS plastic that has not been exposed to light.
Finally, the solution that people recommend, while strenuously insisting it does not imply bleaching activity but actually solubilization of bromine, has all of the characteristics of a peroxide bleach and none of a solubilizing solution. One the one hand, it includes no surfactants and nothing to increase solubility. On the other hand, it requires a bleaching catalyst like UV light (which causes formation of hydroxide radicals, the chromophore-hungry UV degradation products of hydrogen peroxide) and is enhanced by the presence of non-ionic bleach activators like tetraacetylethylenediamine (TAED) from commercial percarbonate or perborate chlorine-free bleaching powders.
None of that would be the case if the objective were simply to grab bromide or bromate off of the surface; you would be better off just washing it off with water with a few cationic surfactants, since even organobromides are soluble and the only major insoluble ionic compound with bromide or bromate is silver bromide. Slap me if I’m wrong, but I really don’t think the ABS on super nintendos is impregnated with silver.
I can only conclude that what’s on the internet about bromine is complete and utter bullshit. Instead, what we’re dealing with here is a peroxide bleaching solution, photocatalyzed by UV light and helped along by TAED bleach activators, that works like a regular peroxide bleach by oxidizing the chromophores. In marketing speak, this mechanism is often referred to as “using active oxygen” by the non-chlorine bleaching powder manufacturers.
The photocatalysis follows the formula
\begin{equation*} H_2 O_2 + h \nu \Rightarrow \bullet OH \end{equation*}where the radical hydroxyl is a highly reactive oxidizing species.
As I thought this, I though that there’s a great way to experimentally determine this - use another bleach. And, voila, someone did, using chlorine as the bleaching agent in the video below. It works just fine on the SNES cover, which is ABS plastic, though since the chlorine is significantly more reactive it did some nasty things to the plastic as well.
https://www.youtube.com/watch?v=9J4yF3OpYZQ
Conclusion: Busted. It’s not the bromine, it’s just normal photodegradation or thermal degradation, and retr0bright and other peroxide-based solutions are just accidental uses of non-chlorine bleach by people who were tinkering.
Basically, the Lego has already been damaged. ABS degradation results in a significant amount of embrittlement and loss of mechanical properties as well as the discoloration. Fortunately, peroxide bleaches are much less strong of an oxidizing agent than chlorine and aren’t likely to further damage the chemical structure of ABS plastic. While it may also start to attack the original dye, the degraded plastic’s yellow pigment is far more vulnerable and will be gone before significant amounts of the original dye disappears.
As an addendum, to make this process even more effective (and since you are in business, the time and space savings are likely to be worth it for you), use a stronger solution of peroxide, add a tiny amount of peroxide bleach activator, and use a dedicated UV light.
Higher concentrations of peroxide can be commercially bought as hair bleaching solutions at concentrations greater than 3%, and if you can buy chemical-grade stuff it’s even better because paying the premium for pharmaceutical grade peroxide isn’t worth it for you; for a non-specialist 10-15% should be a decent balance between safety and effectiveness. Peroxide bleach activators in use worldwide now are primarily TAED and two compounds called NOBS (nonanoyloxybenzene sulfonate) and the related DECOBS, the former of which is prevalent outside of North America and the latter of which is more common inside North America. If you can’t simply buy these in bulk, then use some commercial non-chlorine bleaching powder; the bleaching agents in them are UV-stable and heat-activated, so they won’t contribute much to the bleaching but the bleach activators will still work. Finally, sunlight will work but it’s only the UV component that really matters; higher intensity UV light will lead to higher bleaching activity.
One last thing: the peroxide will gradually lose strength as the UV light bombardment causes it to turn into radical oxidizing agents, which when spent will turn into water. So, replace the solution every once in a while.
[1] Polymer Photodegradation; Page on weathering-symposium.org
[2] Thermal degradation of acrylonitrile-butadiene-styrene (ABS) blends
This is some of the best discussion I’ve found. I’m not sure this part is accurate:
Finally, sunlight will work but it’s only the UV component that really matters; higher intensity UV light will lead to higher bleaching activity.
Shorter wavelengths might matter, but it’s probably not just UV.
To the best of our knowledge, the effect of solar radiation at 45°C on the flow, microstructure and physico-mechanical properties on ABS polymers has not been investigated and the mechanism of yellowing reactions also remains essentially unresolved. The thin degradation layers on specimens are very effective in the loss of mechanical properties, but the reason for these phenomena has not yet been completed.
Butadiene redux
In the process of looking this up, I encountered an even longer and better-sourced Reddit post by u/Grogglebob about the chemistry of retrobriting that also points to butadiene as the culprit. Again, due to the ephemeral nature of Reddit posts I’ll quote it in full:
During the Spring semester of last school year, I was given an assignment to research any topic related to chemistry that I found interesting and give a presentation offering insight into that subject. I have always been fascinated by the process with which old ABS plastics undergo yellowing over years and years of exposure to heat/UV light. On top of that, I am a huge fan of retro video game consoles. That in mind, I decided to dedicate this project to finding out the answer to the question: What is the chemical process that causes this yellowing to occur, and can this process be reversed?
(This is going to be a long one folks, so sorry ahead of time!)
Before I answer the question, lets talk about the Retr0bright formula. The substance contains thickeners for easy application, but the most important compound is the hydrogen peroxide, which is a fairly strong oxidizer. Keep this in mind while reading through my idea.
In order to answer this question, I went to the first place that people would normally go, being Google. I saw that the vast majority of people believed that TBBP-A (Tetrabromobisphenol-A) was the main cause of this yellowing. TBBP-A is a brominated flame-retardant which is roughly thought to react with UV light and oxygen to create a brown compound. I didn’t think that this was correct to being with, since typically elemental bromine has a very dark brown color. In addition to that, the very idea of having only bromides doesn’t work, since you need a cation counterpart to coincide with the bromide anion. It just didn’t seem right.
I decided to go a different route and look at the actual polymer chain itself. The typical retro console plastic is made of a thermoplastic called ABS (Acrylonitrile-butadiene-styrene), which is known to undergo a process called photo-oxidative degradation. This plastic is an amazing polymer blend that takes all of the great things about each of its three constituent parts and mixes them together. One of its more serious downfalls is its tendency to yellow overtime from oxidation (with or without TBBP-A).
While searching the vast literature on the topic, I came across a TON of papers that ran experiments with a whole variety of different conditions while monitoring the photo-oxidative process of ABS plastic samples. Some left the samples outside in a hot climate, while others used a controlled box environment to simulate specific conditions. In the end, they all came to the same obvious conclusion of a horrendously yellow plastic color. At this point, I still had not found any discussion on what chemical process was occurring. I then found an excellent paper that gave fantastic insight into the process.[20]
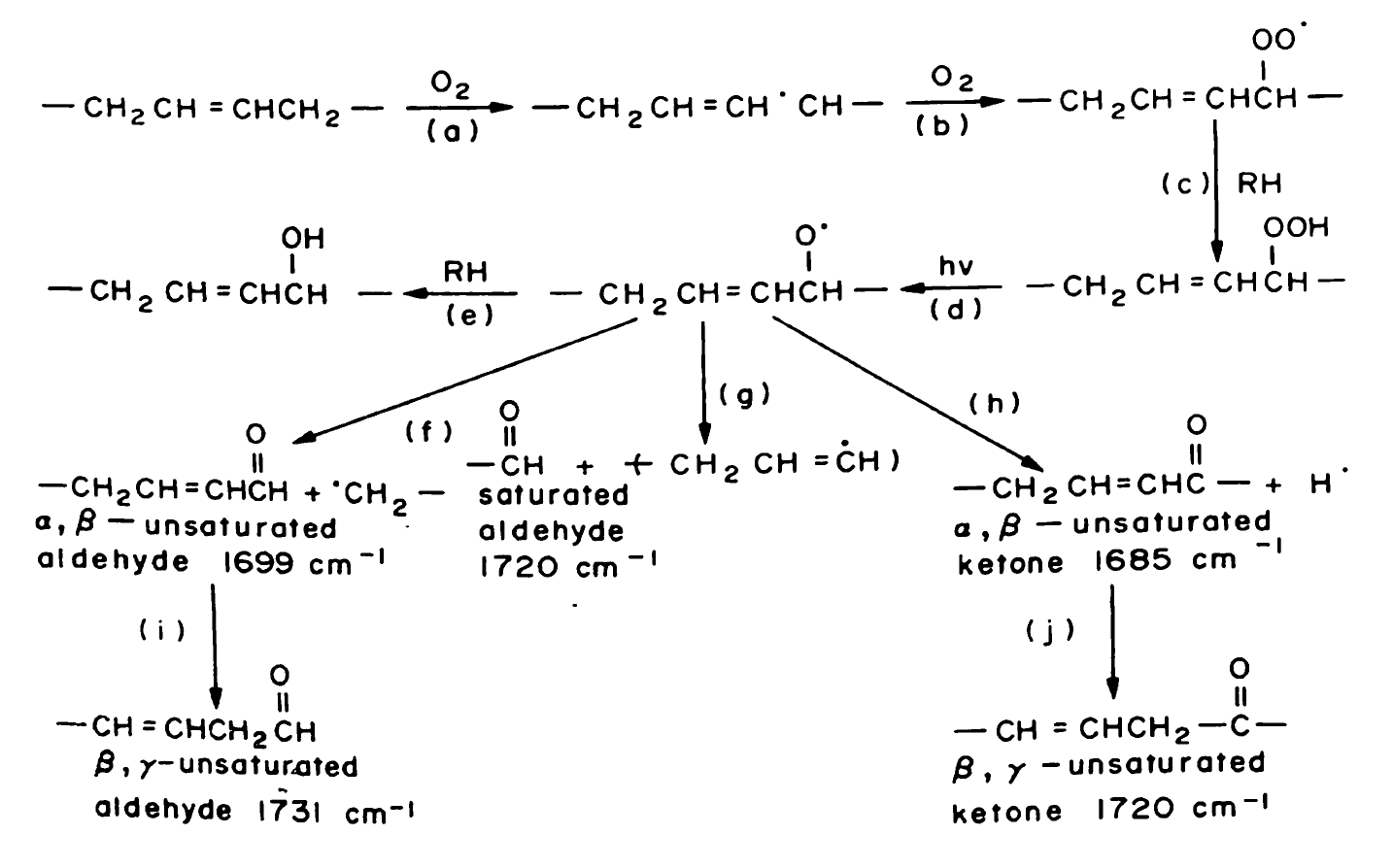
The reaction pathways that these papers proposed seemed perfect in proving what was actually going on here. It appears that as oxygen is exposed to the plastic, the UV light allows for it to react with the carbon-carbon double bonds on the butadiene portion of the polymer chain. After a few reactions, the end product comes out as being a compound group called an α,β-unsaturated carbonyl. In General Chemistry, we are taught that conjugation of chemical compounds causes the lowering of the energy of the emitted light, allowing for light to be emitted in the visible region. The α,β-unsaturated carbonyl has conjugation, so it is not out of the question to assume that this compound causes the yellow color to form.
If we take this compound to be the one that causes the yellowing to occur, then we still need to answer the question as to whether or not this process can be reversed. If we look back at what the butadiene chain looks like in the first place, there are double bonds dotted along the chain allowing for the photo-oxidation to occur. The α,β-unsaturated carbonyl forms on these double bonds, but they themselves contain a double bond. At this point, everything I am saying is pure conjecture, but I feel that there is merit to this idea.
In Retr0bright, I mentioned that hydrogen peroxide is a strong oxidizer, but if you recall, the process which causes yellowing is a photo-oxidative process. I also found an excellent video demonstrating that the presence of a strong oxidizer causes the reverse, where he submerges a yellowed sample in a solution of chlorine and methanol. Naturally, you would expect that the only way to reverse the process would be to use a reducing agent, yet using peroxide and UV light works in reversing the yellowing. I think that the hydrogen peroxide is a strong enough oxidizer to further oxidize the double bond on the α,β-unsaturated carbonyl, thereby removing conjugation and the yellow light emission. If this is the case, then the Retr0bright is not actually reversing the process, but it is instead simply “masking” it.
Well, there is my theory on how Retr0bright really works, so let me know what you think! It seems like a fairly solid idea, but I am definitely open for criticism. Also, I am by no means an expert on this subject, but I will answer your questions as best as I can!
TL;DR
My theory is that Retr0bright does not work in the way that people think. Instead, the process only masks the yellowing by taking oxidation of the plastic one step further. By doing this, the compound causing the yellow is gone but at the cost of (possibly) irreversibly changing the ABS polymer chain. This change could also be attributed to the brittleness of the plastic after treating the plastic with Retr0brite.
EDIT: Added a TL;DR
Project References (sorry for the messiness):
[1] Campbell, Ian M. Introduction to Synthetic Polymers; Oxford University Press: Oxford, 1994.
[2] Strong, Brent A. Plastics: Materials and Processing, 3rd ed.; Pearson Prentice Hall: New Jersey, 2006. pg. 903
[3] Strong, Brent A. Plastics: Materials and Processing, 3rd ed.; Pearson Prentice Hall: New Jersey, 2006. pg. 225
[4] Geek With Envy. https://www.geekwithenvy.com/wp-content/uploads/2013/07/snesYellowing.jpg. (accessed Feb 20, 2016).
[5] Domestic Recycling Symbols. http://members.modernvespa.net/jess/uploads/domestic_recycling_symbols_186.jpg. (accessed Feb 20, 2016).
[6] Acrylonitrile-Butadiene-Styrene Polymers. Encyclopedia of Polymer Science and Technology, 3rd ed.; Wiley-Interscience: New Jersey, 2003; Vol. 1, pp 183-189.
[7] Vintage Computing. http://www.geekwithenvy.com/wp-content/uploads/2013/07/snesYellowing.jpg. (accessed Feb 20, 2016).
[8] Retr0bright. The Retr0bright Project. http://www.retr0bright.com/ (accessed Feb 20, 2016).
[9] myst32YT. Use chemistry to remove the yellow from old plastic. [Not Retr0bright]. YouTube, July 2, 2012. (accessed February 19, 2016).
[10] Acrylonitrile-Butadiene-Styrene Polymers. Encyclopedia of Polymer Science and Technology, 3rd ed.; Wiley-Interscience: New Jersey, 2003; Vol. 1, pp 182.
[11] Comelli, D.; Toja, F.; D’Andrea, C.; Toniolo, L.; Valentini, G.; Lazzari, M.; Nevin, A. Advanced non-invasive fluorescence spectroscopy and imaging for mapping photo-oxidative degradation in acrylonitrile–butadiene–styrene: A study of model samples and of an object from the 1960s. Polym. Degrad. Stab. 2014, 107, 356-365.
[12] Santos, R. M.; Botelho, G. L.; Machado, A. V. Artificial and natural weathering of ABS. Journal of Applied Polymer Science , 2010, 116, 2005-2014.
[13] Bociaga, E.; Trzaskalska, M. Influence of ageing on the gloss, color, and structure of colored ABS. Color Res. Appl. 2015, 00, 00.
[14] Iannuzzi, G.; Mattsson, B.; Rigdahl, M. Color changes due to thermal ageing and artificial weathering of pigmented and textured ABS. Polymer Engineering & Science. 2013, 1687-1695.
[15] Guzel, A., Photo-oxidative Degradation of ABS Copolymer. M.S. Dissertation, Middle East Technical University, Ankara, Turkey, 2009.
[16] Bokria, J. G.; Schlick, S. Spatial effects in the photodegradation of poly(acrylonitrile-butadiene–styrene): a study by ATR-FTIR. Polymer. 2002, 43, 3239-3246.
[17] Salman, R. S.; Al-Shama’a, N. D.; Al-Jarrah, M. F. Effect of Thermal Aging on the Optical Properties of ABS Plastics. Polymer Plastics Technology and Engineering. 1992, 3-4, 213-219.
[18] Shimada, J.; Kabuki, K. The Mechanism of Oxidative Degradation of ABS Resin. Part I. The Mechanism of Thermooxidative Degradation. Journal of Applied Polymer Science. 1968, 12, 655-669.
[19] Shimada, J.; Kabuki, K. The Mechanism of Oxidative Degradation of ABS Resin. Part II. The Mechanism of Photooxidative Degradation. Journal of Applied Polymer Science. 1968, 12, 671-682.
[20] Adeniyi, J. B. Clarification and Discussion of Chemical Transformations Involved in Thermal and Photo-oxidative Degradation of ABS. Eur. Polym. J. 1984, 20, 291-299.
https://www.reddit.com/r/gamecollecting/comments/4pdwhw/my_theory_on_retr0bright/
Here’s the YouTube video u/Grogglebob references:
Use chemistry to remove the yellow from old plastic. [Not Retr0bright]
myst32YT July 2, 2012
https://www.youtube.com/watch?v=9J4yF3OpYZQ
At this point it might be worth mentioning that there is a lot of retrobrite discourse on YouTube.
Retrobrite experiments on YouTube
There’s a lot of retro-computing enthusiasts on YouTube, and so also a lot of retrobrite discourse. Some of it involves demonstrations and experiments on different methods for retrobrite. For example, The 8-Bit Guy (David Murray) has tried a variety of different methods, including:
- Salon creme (hydrogen peroxide cream)
- Hydrogen peroxide solution
- Engine cleaner
- Engine degreaser
- Sodium percarbonate powder in solution
- White vinyl dye
- An ozone generator
The ozone generator particularly caught my eye:
The last thing I’m going to try is my own idea, which is using an ozone generator. Now, the reason I’m trying this is due to an experiment that I did almost a year ago. Now, I didn’t take any video of it. But what I did was I poured some hydrogen peroxide (3%) into a glass container. Then I put small metal pedestal in there and I placed a yellowed key on top of it. Then I covered the top of the container and set it out in the sun. Interestingly enough, the key turned white even though it was never touching the liquid. What this told me was that it was not the liquid itself that was causing the color change. Rather, it was a gas. However, it still required UV light because I tried the experiment again in the dark and it didn’t work. So the question was: did the UV light affect the liquid, creating the required gas? Or did the UV light react with the gas and the plastic directly? Well, that’s what I’m going to try to find out. Now, I’m not a chemist, but after doing some reading on the topic, I thought that maybe one possible result of this combination would be ozone gas. So I bought this ozone generator and I’m going to try it out and see what happens.
[ … ]
And, I have another experiment to show you. During the process of filming this video, I was contacted by a fan of my channel, Richard Blair, who is a professional chemist. And he said he had done some experiments on this and he discovered that heat worked just as well, if not better than UV light. And that’s interesting, because I know the Obsolete Geek and I had a discussion about that just a few months ago and we were wondering what role heat might actually play in the retrobrite process. So, I set out to recreate Richard’s experiment and see how it worked for me. I needed to heat hydrogen peroxide to 150 or 160 degrees Fahrenheit for about 4 hours.
— The 8-Bit Guy, “Adventures in Retrobrite - New techniques for restoring yellowed plastic!” (2:26), Aug. 20, 2017
The full comparison looks like this:
https://youtu.be/qZYbchvSUDY?si=5FfUyNMSv-4kPSaf&t=784
Note that “40% Peroxide” means 40 volume (12% hydrogen peroxide solution).
By the way, I have been saying this is 40% peroxide. But I was recently corrected on this, and apparently 40 volume doesn’t mean 40%. Apparently it’s more like 12% peroxide.
https://youtu.be/qZYbchvSUDY?si=5FfUyNMSv-4kPSaf&t=1008
Lab-grade hydrogen peroxide is 30% by weight, the kind used in piranha cleaning. At concentrations of 40% hydrogen peroxide is considered hazardous waste, and higher concentrations like 70-98% are used as rocket propellent.
Here’s what I found particularly interesting: normally the process is done underwater, in the absence of oxygen gas (except for whatever’s dissolved in the water). But apparently it also works in air:
Interestingly enough, the key turned white even though it was never touching the liquid.
and in ozone:
Both keys spent 24 hours in a bag filled with ozone, but only the 7 key got UV light along with it. In fact, comparing with the control, you can see that ozone by itself is apparently useless. But ozone with UV seems to work. However, if you compare with the key from the salon cream, you can see that ozone wasn’t as effective. I imagine that ozone would probably completely restore the plastic it just probably needs more than 4 hours.
https://www.youtube.com/watch?v=qZYbchvSUDY&t=644
It’s possible that it was a gas causing the change, but it’s potentially also just temperature, right?
One criticism of the experimental design: it should have used other controls, such as:
- just heat
- just UV
- just water
- heat and UV
- water and UV
- heat and water
- heat, UV, and water
It’s also weird that engine cleaner wasn’t used with UV.
The community has come up with a process of removing yellowing from plastics using peroxide. It is typically referred to as Retrobrite. Normally, you need to apply the peroxide cream and put the item out in the sun until the yellowing goes away. This has potential of going too far and damaging the plastic resulting in marbling and streaks.
Recently, people have started looking into ways to control the process by performing it indoors using heat, UV light and/or ozone generators.
I tried a method that doesn’t involve UV, heat or ozone. Does it work?
— Adrian’s Digital Basement, “A new way to fix yellowed plastics indoors?”
“Can you retrobrite under blue LED light? The answer, at least in this case, was yes. People are saying you need ozone, or you need heat, or you need UV. Well, 457 nanometers, which is the royal blue LEDs used in this is not UV, and yet it absolutely retrobrited this mouse perfectly. But anyhow, I will do further experiments and probably create another video to update you guys.”
Embrittlement and more complicating factors
So there’s a lot of guesswork going on without a firm understanding of the fundamental mechanism. For example, there is no agreement about:
What polymers the treatment works on.
Is it just ABS or others, too?
What the actual reaction is.
Is it the flame-retardant bromide additives or is it just ABS oxidation?
Surely there is some bromide-free ABS to test.
Why it requires both peroxide and UV.
Presumably heat would work just as well.
What wavelength is the most effective.
Is it UVA? Blue light? Something else?
How much temperature affects the process.
How much does a higher temperature accelerate the de-yellowing process?
How deep into the plastic surface does the yellowing occur?
Presumably it’s just a thin layer of the surface, but is it just a few microns or is it more than a hundred microns? If it’s just a few microns, then wouldn’t chemically removing a thin layer of the surface have the same effect?
More concerning is there is little or no discussion about:
- How long the treatment reverses the yellowing (months? years?).
- Whether the process makes the plastics more or less brittle.
I have found little discussion about whether retro-brite processes make ABS more brittle, and no data at all. I’m guessing this is because measuring brittleness of plastic is difficult, especially in a non-destructive fashion.
This article shows evidence that yellowing after Retr0Brighting can indeed return over time. The suprise is it happens with seemingly very little UV light. What’s more, it only appears to be the plastic initially damaged by yellowing which re-yellows. Plastic that was originally unaffected stays that way.
[ … ]
The original damage from light causes degraded or free bromides throughout the case from the fire retardants. Retr0Bright only takes these away from the surface layer. However these bromides can migrate fairly freely through the polymer. They don’t need light to do this. Migration is probably accelerated in hot conditions. These pre-existing bromines from the original damage migrate to the top and within a few years the surface is yellowed again. The rate of the regression depends on the inital extent of yellowing (the more yellowed, the more bromides down deep), heat exposure and the nature/quality of the plastics.
https://www.classic-computers.org.nz/blog/2013-01-15-retr0bright-only-temporary.htm
Unsatisfied with Nintendo’s answer, I decided to dig deeper. I contacted Dr. Rudolph D. Deanin, founder of the graduate program in Plastics Engineering at the University of Massachusetts, Lowell and director of the program for over thirty years. I told him everything I know about plastic chassis discoloration in both the Super Nintendo and Macintosh computers and asked what he thought might be causing it.
“The plastics most commonly used to make the structural cases for electronic equipment are polypropylene, impact styrene, and ABS,” replied Deanin. “These all tend to discolor and embrittle gradually when exposed to UV and/or heat. They become oxidized and develop conjugated unsaturation, which produces color. They crosslink or degrade, which causes brittleness.”
From looking at a stamp on the Super Nintendo’s plastic case, I learned that the case is composed of ABS, which is a rugged, durable plastic that is sadly more susceptible to discoloration and degradation from both UV and heat than the alternatives.
“There are other plastics which would be more stable,” Deanin continued, “but manufacturers avoid them because they are more expensive and/or more difficult to process.”
Instead of using more expensive plastics, manufactures put additives known as stabilizers, absorbers, or blockers into the plastic mixture to reduce the effects of degradation. They also get creative with their use of pigmentation.
“Since most discoloration is toward yellowing, some manufacturers add a little blue to neutralize the yellow,” Deanin said. “This gives a temporary reprieve, but eventually the yellow keeps growing and overpowers it anyway.”
[ … ]
Despite numerous demonstrations of this technique and a seemingly sound theory behind it, the jury is still out on whether this method harms plastics or color pigmentation in the long-term. Be that as it may, this technique is currently the best, least-destructive method we have of reducing yellowing in ABS plastics.
http://www.vintagecomputing.com/index.php/archives/189
It’s also manifestly untrue that there have been no experimental studies on polymer yellowing caused by exposure to light. However, many of the experiments have poor controls. To quote one of the books referenced in the Quora post:
In spite of extensive studies of photo degradation of polymers, there are many unsolved problems. Many published results are even contradictory and it is difficult to compare them, probably because results are obtained on different samples of the same polymer and under different experimental conditions. Many proposed mechanisms are still not experimentally proven. However, the total collected knowledge in published papers gives a general image of physical processes and chemical reactions occurring during photo- degradation and photoageing of polymers.
[ … ]
The photochemical transformation of ABS in the presence of oxygen involves reactions in the polybutadiene nodules and in the styrene-acrylonitrile macrophase [1114]. The photo-oxidation of the polybutadiene nodules is responsible for the loss of the mechanical properties of ABS, and also causes an induced oxidation of the polystyrene component in the styrene-acrylonitrile macro phase. The main effect of this reaction is the formation of highly absorbing photo-oxidation products that are responsible for the discoloration of ABS on irradiation at long wavelengths in the presence of oxygen.
https://books.google.com/books?hl=en&lr=&id=0MTrCAAAQBAJ&oi=fnd&pg=PR15
For the case of ABS, here’s two papers cited:
This experimental observation could result of (1) a control of the oxidation kinetics by the diffusion of atmospheric oxygen into the polymer film, ( 2 ) a decrease of the absorbed light intensity through the film that would result of an important initial absorption. This last effect would be enhanced if the polymer developed an important increase of absorption in the UV and the visible range (“yellowing”).
[ … ]
The evolution of the photochemical degradation of ABS has been studied in conditions of long wavelength irradiation (λ’s> 300 nm). The main photoproducts involved in the oxidative evolution have been identified by using FTIR spectroscopy and chemical titrations. A particular attention has been devoted to α–β unsaturated ketones that appear as secondary photoproducts. Those ketones present a low photochemical stability when exposed in the range 300–400 nm. Conditions for their formation have been experimentally studied. Formation of oxidation photoproducts has been also studied at the macroscopic level and it has been shown that their repartition in the polymer is heterogeneous. The origins of the heterogenities have been studied.
[ … ]
It must be pointed out that photooxidation of ABS leads to a large increase in the UV and visible absorption and consequently to a pronounced yellowing of the material (this behavior will be described in a forthcoming paper).
[ … ]
Another effect is suspected and it relates to the control of kinetics by the diffusion of oxygen into the polymer sample. This effect is in fact clearly proved by analysis of oxidation photoproducts profiles: investigations through the film show that photoproducts are localized essentially in the surface layers while the core of the film appears to remain
https://doi.org/10.1002/pola.1991.080290510
Photo-oxidation of ABS: Part 2—Origin of the photodiscoloration on irradiation at long wavelengths
- Jouan, J.L. Gardette
[ … ]
The reactions that lead to the discoloration of acrylonitrile-butadiene-styrene terpolymers (ABS) on exposure to long-wavelength irradiation (λ > 300 nm) in the presence of oxygen have been studied. The photoproducts responsible for the discoloration are obtained by photo-oxidation of the polystyrene component of the styrene-acrylonitrile (SAN) macrophase. It is shown that the polystyrene oxidation is mainly initiated by radical species obtained in photo-oxidation of the polybutadiene nodules. The considerable photo-oxidability of polybutadiene accounts for the high rate of discoloration of ABS in comparison with the discoloration of polystyrene homopolymer. In conditions of accelerated artificial photo-ageing, oxygen starvation effects on the photodiscoloration of ABS are observed. Similar effects have been observed formerly in the photo-oxidation of the polybutadiene nodules.
[ … ]
CONCLUSION
The photochemical transformation of ABS in the presence of oxygen involves reactions in the polybutadiene nodules and the SAN macrophase. The photo-oxidation of the polybutadiene nodules is revealed by the changes in the IR spectra of the photo-oxidized samples. The oxidation of the polybutadiene nodules is ultimately responsible for the detrimental effect of irradiation on the mechanical properties of ABS. The second consequence of the oxidation of the polybutadiene nodules is an induced oxidation of the polystyrene component in the SAN macrophase. The main effect of this reaction is the formation of highly absorbing photo-oxidation products that are responsible for the discoloration of ABS on irradiation in the presence of oxygen.
https://doi.org/10.1016/0141-3910(92)90054-9
In a primary photodissociation of phthalate groups, the radicals formed react in the “cage” after a fast decarboxylation or decarbonylation ; this cage reaction accounts for the photolysis products. The formation of products on the styrene unit can be induced too by the radicals escaping the cage. In the presence of oxygen, the radicals can be oxidized out of the cage or can be competitively trapped by the styrene units. Direct photooxidation of the phthalate groups or induced oxidation of the styrene units can be observed. Moreover, the products formed in photolytic conditions can be photooxidized. This mechanism can be ascertained by the introduction of antioxidant redox in the matrix. Such an antioxidant inhibits the induced photooxidation without inhibiting the direct photooxidations.
https://doi.org/10.1002/masy.19890250124
There’s also this paper cited in the Wikipedia article on retrobright that mentions the mentions the chemistry of polymer yellowing is “still very controversial”:
Photodegradation of polystyrene in air causes discoloration (yellowing), cross-linking, and chain scission due to oxidation. A photodegradation process was also proposed on the basis of the IR spectrum of the photoirradiated film, which indicated the formation of peroxy radical and hydroperoxide intermediates. The photochemical reactions cause the dissociation of a polystyryl radical by creating an electrochemical excited state. The polystyryl radical is converted to peroxy radicals by reacting with oxygen. The peroxy radical undergoes chain scission and formation of carbonyl compounds.
[ … ]
The main reactions observed are bond session, chain crosslinking and oxidative degradation. In spite of these great efforts the mode of photodegradation of polystyrene is still very controversial.
— Emad Yousif & Raghad Haddad. “Photodegradation and photostabilization of polymers, especially polystyrene: review” (2013)
https://doi.org/10.1186/2193-1801-2-398
To complicate matters even further, there’s an older paper cited in a forum post cited by the Wikipedia page that suggests ABS yellowing occurs when the polymer is left in the dark (!), not when it is exposed to light:
The commonly observed effect of weathered polymer samples continuing to change color in the dark was studied in some detail for polycarbonate, styrene–acrylonitrile copolymer (SAN), and various combinations with SAN–polybutadiene rubber graft (SRG) copolymer. All of the formulations became more yellow upon storage in the dark after accelerated weathering, and all behaved very similarly. There was a fairly rapid shift of 1–2b units (as defined in CIELAB color space) over the first two weeks followed by a slower, linear shift that continued for many months. This amount of color shift is enough to be visible to the eye in side-by-side comparisons. Most of the additional color generated in the dark could be bleached by exposing the samples to sunlight through a south-facing window. The samples usually reached the original color after 2–4 days of window exposure. Putting the samples back in the dark caused the yellowing to start up again, although the rapid yellowing often was not as much as the first time. Samples aged under natural conditions outdoors also underwent a small color shift upon storage for 5 months and substantial color shifts after two years.
[ … ]
Many people involved with accelerated weathering of polymers are familiar with the phenomenon of post-exposure yellowing, sometimes called post-actinic yellowing or the “drawer effect”. Some samples removed from an accelerated weathering tester have been found to continue to change color at room temperature in the dark. Although this effect has been casually observed by many, there are only a few reports of it in the literature. French workers have observed reversible dark color shifts for acrylonitrile–butadiene–styrene polymers (ABS) […] In this report we seek no chemical explanation for the phenomena observed in aromatic engineering thermoplastics, but only describe the rate, magnitude, and reversibility upon exposure to sunlight through glass.
— James E. Pickett, “Reversible post-exposure yellowing of weathered polymers” (2004)
https://doi.org/10.1016/j.polymdegradstab.2004.03.008
There’s even a recent review paper on polymer yellowing:
[ … ] one of the most important facets giving significant concern to the polymer industry is that of discolouration or more specifically the “Yellowing” phenomenon. Although the literature is abundant covering various aspects of this problem it can be what one may describe as a serious “bug” in the progress of many polymer applications and indeed can limit their acceptance in many products. These may be in clear films and bottles, white goods or recycling and post-use where heat and light can discolour polymer materials. The science underpinning the yellowing can be due to the additives, more importantly, the phenolic/amine antioxidants and/or inherent in the polymers themselves especially polyaromatics such as polyesters or polycarbonates, epoxy-amines, and polystyrene to name but a few. In many industrial applications solving or overcoming this issue is not always easy and can be problematic. The chemistry of these processes will be discussed and highlighted and effective modes of stabilisation and/or inhibition presented with often unconventional detailed examples and their structural developments for optimisation of additive performance.
— Norman S. Allen, Michele Edge, Sajid Hussain. “Perspectives on yellowing in the degradation of polymer materials: inter-relationship of structure, mechanisms and modes of stabilisation” (2022)
Summary & takeaways
Here’s my takeaway: the yellowing mechanism is initiated by the polybutadiene component of the ABS, which is less stable than the other components. However, the polystyrene is the component that does the actual yellowing.
If I had to guess, I think the plastic yellowing reaction looks more like this:
polystyrene + O2 + hν → P1/P2
where hν is a photon of suitable wavelength and P1/P2 are hydroperoxide photoproducts. If so, the “retrobrite” reaction looks more like this, where the peroxide acts as a reducing agent that pulls the oxygen out of the photoproducts:
P1/P2 + 2 H2O2 + hν → polystyrene + 2 O2 + 2 H2O
This would explain the bubbles produced, assuming they are actually oxygen, which could be determined by a flame test. Presumably heat could also take the place of hν to accelerate the reaction, although black-body radiation at 160°F (344 K) is almost all in the long infrared range, much lower than a photon of UV or blue light. Perhaps a 940 nm infrared LED could be used for retrobrite? It would be interesting to see if UV light can be used to retrobrite at lower temperatures such as 50°F (10°C) as this would help establish to what extent heat or light is necessary for the reaction to proceed.
Footnotes
| [1] | As explained on retr0bright.com:
Heat may be used as a substitute for UV exposure, submerging ABS plastic in 40 unit (approx 12%) hydrogen peroxide solution at 71°C (or around 160°F) has been shown to be effective. https://en.wikipedia.org/wiki/Retr0bright
https://www.youtube.com/watch?v=jdWRsjnVD3s Adventures in Retrobrite - New techniques for restoring yellowed plastic! |
